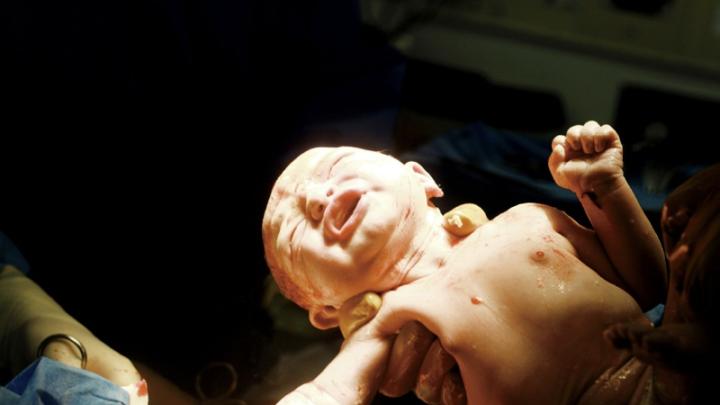
In May 2003 came the joyous birth of Prairie Cummings Resch, first child of Zoe Cummings Resch ’92. All had gone according to plan: Resch lay down on a surgical table. An anesthesiologist inserted an analgesic into her spine, and she became impervious to pain below her waist. The obstetrician pressed a No. 10 blade into Resch’s lower abdomen, and made a six-inch horizontal cut. The doctor divided the skin, stanched blood, and, reaching Resch’s large abdominal muscle, parted it. He slipped his knife through the opening, and cut into the peritoneum, the thin membrane that lines the abdominal cavity. He sliced into Resch’s uterus. A medical resident reached in and pulled Prairie out feet first; this baby was in breech position, upside down in the womb.
Resch felt “a lot of rough pushing and pulling,” a “painless suction sensation,” as if her body were “a tar pit the baby was wrested from.” She heard the doctor say to the resident: “Hold her up by the hips,” and Resch peered down. She saw her daughter for the first time, wet and squirming. Prairie wailed. Resch’s husband held the baby next to Resch’s cheek. Resch felt “overwhelmed by emotions”—“joy, awe, anxiety, relief, surprise.” She gave thanks for her healthy baby, and for modern obstetrical care.
In the next six years, Resch would have two more babies—each by C-section, despite uncomplicated pregnancies. She says she doesn’t regret any of these surgeries: she has three healthy children and each surgery “went well.” But her story and those of a number of other women shed light on why one-third of American babies now enter the world via the knife, in operating rooms, ringed by technicians. In 1970, only 5 percent of American children were born this way.
Obstetrics in modern America is a contentious subject in general. Birth and the actions surrounding it—medical and otherwise—evoke strong emotions. The discussion is often framed ideologically as a matter of nature versus technology and which side knows best, or in stark political and economic terms as a contest of power and money. The issue of C-sections, in particular, is much contested.
It’s useful to see cesareans’ ascendance as a result of the ways doctors, patients, and hospitals perceive and react to risk—and of how medicine has developed in this context. Understanding such interactive reasons, and responding thoughtfully to them, experts say, could help reduce the procedure’s use.
In 1985, amid increasing disparity among nations in the number of cesarean births, the World Health Organization (WHO) set out to determine an optimal rate. After reviewing the percentage of pregnancies with complications best resolved by C-section, WHO announced that a cesarean rate of 15 percent was ideal—about one-half the current U.S. rate. The 15 percent rate, WHO reasoned, would optimally prevent childbirth injuries and deaths, but many women and babies would avoid unnecessary and potentially harmful surgery. WHO has since modified this specific recommendation, stating in 2009 that “the optimum rate is unknown,” but that “both very low and very high rates of cesarean section can be dangerous.”
Most U.S. experts—whether high-risk obstetricians or home-birth midwives—agree that the U.S. rate is higher than medically necessary and acknowledge that many women are undergoing major surgery for avoidable reasons. Jeffrey Ecker, M.D. ’88, professor of obstetrics, gynecology, and reproductive biology, is a high-risk obstetrician at Massachusetts General Hospital and director of his department’s quality and safety program. A few years ago, working with the Massachusetts Department of Public Health, he compared C-section rates among the commonwealth’s hospitals. The study, like similar ones in other states, found great disparities: Massachusetts hospitals showed as much as a threefold variation in frequency of cesareans. These disparate rates, Ecker says, “can’t all be optimal,” and, he adds, it’s “certainly very difficult to demonstrate that higher cesarean rates are associated with better outcomes.”
Demographic changes and shifts in maternal health may have contributed to the rise in the use of C-sections in recent decades. Pregnant women, overall, have become older and heavier, and older, heavier women undergo more C-sections. But such factors don’t account for all the differences shown in Ecker’s study, which sought to control for them by looking only at pregnancies that had progressed well: in which fetuses had reached full term, were normal weights, and in which labor had begun spontaneously (i.e., doctors had found no reason to induce labor early). Even among these cases, hospitals varied significantly in the frequency of C-sections.
Such disparities matter because cesareans are expensive—on average, a cesarean costs about $20,000, a vaginal birth about $11,500—and also carry significant risks. When compared with vaginal birth, cesarean delivery increases low-risk women’s chances of certain rare but potentially life-threatening problems, such as hemorrhage, blood clots, and bowel obstruction. More frequent risks include bladder damage, infection, and enduring pain. Women who’ve delivered by C-section face greater likelihood of future complications in pregnancy, including uterine rupture or conditions in which the placenta covers the opening to the cervix (placenta previa), adheres abnormally to the uterine wall (placenta accreta), or separates from it (placenta abruption). These women are also less likely to breast-feed, and may be at greater risk for depression and post-traumatic stress.
Babies face risks, too: they may be cut, or asphyxiate if the medical team has difficulty pulling them out. Those born by cesarean are more likely to experience respiratory distress and, later, to have asthma; controlled studies have found increased rates of obesity among American babies born this way. The fetus of a mother who’s already had one cesarean also seems to be at increased risk because it faces greater danger when growing in a uterus with a surgical scar.
Proponents of more-natural birth argue, too, that vaginal birth facilitates quicker, perhaps better, bonding with newborns. Babies born vaginally receive a coating of immune-boosting microbes, and their intestines are more likely to have early colonization with beneficial bacteria—protections that babies delivered surgically miss out on. A bacterial deficit in babies’ guts, some scientists speculate, may even be the factor that accounts for the higher obesity rates among those born by cesarean.
A C-section likely saved the life of Alexandra Houck ’87, a family practitioner with Harvard University Health Services. Late in her first pregnancy, Houck developed vaginal bleeding. At first her obstetrician hoped the blood was a sign of early labor. But when the doctor saw the extent of the flow, she feared that Houck’s placenta had separated from the uterine wall—a placental abruption, which can cut blood flow to the baby, and cause hemorrhaging in the mother. The physician ordered an emergency C-section, which confirmed her fears: Houck did have placental abruption. “Potentially,” Houck says, “before we had modern medicine, somebody in my position would have died, and the baby would have died.”
Many cesareans happen, though, for reasons more complex. During her first pregnancy, Zoe Resch had hoped and prepared for natural childbirth; with her husband, she had attended natural birthing classes, learned breathing exercises, practiced with a birthing ball—and written up a “birth plan” that included trying to go without pain relief and, generally, avoiding as many medical interventions as possible. She was low-risk: 32 years old, without diabetes, high blood pressure, or other medical problems that increase risks in pregnancy and labor. Then, toward the end of her uneventful pregnancy, her obstetrician discovered the baby’s breech presentation. Several times in the next weeks, the physician attempted an “external version”—using her hands and pushing carefully on Resch’s abdomen, she tried to turn Prairie head down. But Resch felt only her daughter’s fierce kicks in response, and heard the baby’s heart on a monitor, thumping faster. Prairie, Resch says, “was having none of it.” The baby remained head up, hind end lodged in her mother’s pelvis.
Though rare—about 3 percent of babies end up in breech position—upside-down birth complicates delivery. Breech babies are more likely to get stuck during birth and, Ecker says, there is a 1 percent to 3 percent chance of injury to the baby during a vaginal delivery with an experienced provider. Because there’s a calculable risk, and C-sections are available, doctors have come to shy away from vaginal breech deliveries. The year before Prairie was born, the American Congress of Obstetricians and Gynecologists (ACOG) formally recommended scheduled cesareans for breeches. Resch remembers no discussion with her doctor about the possibility of a vaginal delivery for Prairie.
But in 2006, after Prairie’s birth, ACOG revised its position, stating that a clinician with sufficient experience and support might appropriately assist in vaginal breech deliveries. Yet such experienced obstetricians are ever harder to find. In a dynamic that is repeated in other medical care, doctors perform cesareans, in part, because they aren’t trained to favor or perform less-invasive techniques. With inadequate training and experience, liabilities and patients’ risk increase. Thus, few hospitals even offer the option of vaginal breech delivery. “It’s like all practice in medicine,” Ecker says. “What you become used to becomes the standard. And what hasn’t been done becomes more difficult to offer.”
Perceptions of risk—on the part of mothers, doctors, and hospital administrators—explain much of the dynamic that has raised the C-section rate, Ecker says. Risk perception and tolerance help determine professional standards of care, influence hospital protocols, mold the media’s telling of stories, and even influence laws. All these forces interact in complex ways. Talking about the cesarean rate, therefore, is different from talking about, attending, or—in the case of the mother herself—living a particular pregnancy and labor. Saying that a certain percentage of C-sections are unnecessary is fairly simple. But weighing risks and knowing whether surgery is necessary in a particular case—or even whether a surgery was necessary in retrospect—is much more complex, and fraught with emotion. The obstetrician sees C-sections as generally safe, and if the outcome he or she wants to avoid is dire, even devastating—such as a baby’s becoming stuck and deprived of oxygen, which could lead to cerebral palsy—why wait to find out what will happen, however unlikely that outcome may be?
The legal climate reinforces this dynamic. “No one gets sued for doing a C-section,” obstetricians famously say. They do get sued, Ecker says, for not intervening. Michelle Mello, professor of law and public health at Harvard’s School of Public Health, studies malpractice law and medical injury. Her study of states with limitations on doctors’ liability found that they have lower C-section rates. But the finding was not “huge,” she says. She believes that such studies “may not entirely capture the effect of liability pressure on C-section rates” because they “don’t compare to a world where there is no liability fear.”
Doctors, Mello says, tend to overestimate their liability risk: “Regardless of whether, from a scholarly perspective, they’re in a ‘low risk’ or ‘high risk’ environment, they all feel like they’re at high risk.” Skewed perception of risk, she says, drives defensive decisions.
Obstetric patients, of course, also have trouble with risk perception. Again, the difficulty arises in part because the worst outcomes—like cerebral palsy and infants’ deaths—although rare, are distressing and easily remembered, and so shape patients’ decisions. Imagine the challenge of risk perception if, when pregnant with your first child, you were researching adverse childbirth outcomes as part of your job. This was Mello’s situation in 2008. Cesarean “wasn’t what I was hoping for,” she says. But after 27 hours of labor, she says, her situation was one “that was very common in the catastrophic injury cases” she’d been studying: “prolonged second-stage labor [the “pushing” stage] and fetal distress.” She was not “about to start arguing” with her provider “about appropriate course of action.” She was wheeled to the operating room.
For Mello’s second pregnancy, her obstetrician “laid out all the risks,” and the researcher found herself weighing the dangers in typically human ways—by looking not just at data, but at “the anecdotes that weigh on you,” she recalls. “We know that people evaluate risk not just on their understanding of statistical information, but on ‘what happened to my friend,’ or ‘what happened to my sister,’” She and her husband “came in with a few of those [stories], too.” She chose a scheduled cesarean.
Ecker, the high-risk obstetrician, says that his patients, too, are influenced by tales—the tragedy on television, the co-worker’s near-miss, the warning online. Patients often focus, Ecker says, on the numerator (the very rare cases) rather than the denominator (the great majority for whom everything goes well).
According to the U.S. Department of Health and Human Services, 85 percent of American pregnancies achieve full term without complications. That’s a figure that many midwives—who in 2011 attended 11 percent of births, mostly as staff in hospitals—believe Americans often lose sight of. They say that simply seeing pregnancy and birth as normal, rather than a priori as a medical problem, would help lower the C-section rate. Cara Osborne, S.D. ’07, a certified nurse midwife and professor at the Eleanor Mann School of Nursing at the University of Arkansas, believes that “an atmosphere of fear” surrounding childbirth drives C-sections. Midwives see birth, she says, as a well-evolved, physiologically sound process that, with the right support, usually turns out fine. (Otherwise, she says, the human species wouldn’t have a population problem.) Of course childbirth is “an inherently risky endeavor,” she says. “But we’ve somehow gotten to a place where we’ve lost trust in physiology.”
Midwives’ training, Osborne says, focuses on optimizing the chances of vaginal birth throughout prenatal care and labor. Midwives usually spend more time with laboring women than obstetricians do, and studies have shown that even passive, nonmedical support during labor leads to better birth outcomes. Midwives are also far more tolerant of slow labors, and are therefore less likely to determine “failure to progress” (when a provider decides that labor is proceeding too slowly to be safe) or “obstructed labor” (caused by a mismatch, of position or size, between the baby’s head and mother’s pelvis)—among the more commonly stated reasons for proceeding to cesareans, says Janet Singer ’84. A midwife who teaches medical students and residents at Women & Infants Hospital of Rhode Island in Providence, Singer adds that “failure to progress” is perhaps the most-preventable reason for cesareans. In the twentieth century, diagnoses of failure to progress rose along with the C-section rate: from 3.8 percent in 1970 to 11.6 percent in 1989 to 16.1 percent by 1995, according to a 2000 study in Obstetrics and Gynecology.
Generally, Singer says, midwives are more “invested” in vaginal deliveries by virtue of training and mind-set—and because of this investment, more likely to help women give birth vaginally.
In some parts of the world, of course, childbirth risks stem from causes that apply less in developed economies. In Haiti, for example, where women’s underlying health and access to care is poor, the lifetime risk of dying in childbirth is one in 40. “That’s totally unacceptable,” says Osborne, who trains Haitians as lay midwives. In Haiti and other poor countries, Osborne and other experts say, women need access to more interventions, including C-sections, but in the United States, she adds, “we are making the risk [of childbirth] higher than it needs to be by interfering with the physiologic process.” Generally, she says, “if we could let nature take its course, we would be in a much better position. But that’s just not the prevailing culture.”
To illustrate the difference in midwives’ point of view, Singer tells of meeting an anesthesiologist who had never collaborated with a midwife. He asked about her work. “In a nutshell,” Singer said to him, “we don’t see birth as a disaster waiting to happen.” “But,” the anesthesiologist replied, “birth is a disaster waiting to happen.”
Ecker says that his study of Massachusetts hospitals found that those with “midwives practicing at them seemed to have lower C-section rates.” But, he asks rhetorically, “is that because somehow midwives attract a population that’s at lower risk? Or is it because midwives and their style of care permeate an institution?” It’s difficult, he says, to tease out the answer.
What is clear is that initial cesareans drive subsequent ones. A woman who has had a cesarean has a 90 percent chance of giving birth by C-section again. Because the procedure increases a woman’s statistical risk of complications in future pregnancies and labors, doctors are more inclined to schedule C-sections or arrange emergency surgery at the first sign of trouble. Zoe Resch decided, in her second pregnancy, to try for a “VBAC”—a vaginal birth after cesarean. But because she’d had a C-section, her doctor, “just as a matter of routine,” she says, scheduled a cesarean for Resch’s due date, in case plans for vaginal birth went awry. Meanwhile, Resch had learned that she was carrying a large (statistically more difficult to deliver) baby. By the end of her pregnancy, she felt exhausted by pre-labor contractions, even as she cared for her toddler at home. She hadn’t gone into active labor, but drove to the hospital on the day of her scheduled C-section. Once at the hospital, with no pressure from her doctor, Resch says, she opted to go ahead with the scheduled surgery.
“Trial of labor”—attempting a vaginal birth after a prior C-section—has become rare, and successful ones—VBACs—even rarer. “We know from studies that what increases your chances of having a VBAC are things like not having epidural anesthesia, and being up and moving about, and having continuous labor support,” Cara Osborne says. “But because, in many practitioners’ minds, the trial of labor is unlikely to work, they’re setting up for a surgical scenario.” Hospitals may encourage or even require a woman to use epidural anesthesia during labor, she explains, “because they want to have it on board if she has a C-section.” In addition, hospitals usually require continuous fetal monitoring in the form of wires attached to the laboring woman’s abdomen, which restricts mobility. As Osborne points out, “The things that would help someone have a successful VBAC are often things that are not offered to her, because the assumption is that this is liable to end in another C-section.”
Once Resch had had two C-sections, giving birth vaginally to her third baby was not an option at her hospital. Studies have shown that the risks of vaginal birth after two C-sections exceed the risks of third cesareans—although some hospitals do allow trials of labor in certain cases.
Because subsequent C-sections are so common, Ecker and others say that minimizing unnecessary first cesareans is crucial to reducing the rate overall. Massachusetts hospitals that have higher VBAC rates, Ecker says, also have lower rates of first C-sections. “So it argues,” he says, “that there’s something in the [hospital’s] culture or process of care that’s making a difference.” Again, “figuring out what that is,” he says, “is really difficult.”
One clue may lie in what some experts call “cascading interventions”—medical actions that lead to other medical actions that evolve into more invasive steps, including C-sections. Inducing labor, for example—in which a provider tries to stimulate a pregnant woman’s contractions through synthetic hormones or by stripping part of the membrane from her uterine wall—has been found to increase the likelihood of cesareans in first-time mothers.
Continuous electronic fetal monitoring (CFM), which tracks a baby’s heart rate throughout labor, is also associated with higher cesarean rates. “It was hypothesized,” Ecker explains, “that [CFM, developed in the late 1960s] would reduce rates of cerebral palsy.” Based on this hypothesis, the technology became widely used. In the great majority of U.S. hospitals, CFM is standard care; a 2005 study found that 87 percent of laboring American women were attached to monitors most or all of the time. Meanwhile, Ecker adds, studies found that CFM had not reduced the incidence of cerebral palsy. But CFM did seem to increase C-section rates, he says: doctors were “seeing these wiggles and squiggles”—changes in fetal heart rate—“that they weren’t seeing before.” They would get nervous and conclude, “We’ve got to do something about it. Let’s do a C-section.”
Now that physicians are coming to understand the process, the solution is not, Ecker says, to avoid monitoring altogether. Listening intermittently with a handheld device catches problems without producing phantom ones. Nevertheless, he notes that “there are a bunch of reasons why [continuous monitoring] isn’t going away. It’s built into the structure of the care we provide.”
This “structure of care”—the patterned way in which obstetrics happens—builds in more common interventions that may lead to others. Professor of obstetrics, gynecology, and reproductive biology Ellice Lieberman led a crucial study in 2005 that showed that epidural use increased the likelihood of an abnormally positioned baby at the time of delivery. Doctors already knew that the anesthetic made fever in labor more likely and tended to prolong labors, and knew that women with babies in an abnormal position called “occiput posterior” were more likely to receive an epidural. Lieberman’s study showed that “it’s not that women are coming in and getting epidurals because their baby’s in an abnormal position,” she says. Rather, babies were in occiput posterior position, in some cases, because of the epidural (as yet, no one knows why). The study found the position four times as often in women who used epidurals as in those who didn’t—but no significant difference in frequency of abnormal position before the women had chosen the anesthesia. The treatment engendered the medical situation.
Abnormal position often leads to a diagnosis of “failure to progress,” which leads to some form of operative delivery—cesarean or the use of vacuum and/or forceps. As with breech deliveries, doctors usually choose C-section. Use of forceps or vacuum is less invasive, but has its own risks. And once again, obstetricians have become less familiar with using these procedures as C-sections have become the norm.
After rising steadily for years, in 2011 the U.S. C-section rate remained virtually unchanged from 2010. Perhaps it has stabilized, or is even about to head down. Such a dip happened once before: between 1990 and 1996, amid concern about unnecessary C-sections, cesarean surgeries in the United States declined by a few percentage points. Then the rate rose again.
In the early to mid twentieth century, doctors sought, and pregnant women demanded, more interventions in childbirth—to relieve pain and prevent injury and death. Partly as a result, by the 1960s women commonly labored alone, confined to hospital beds and under sedation. Forceps deliveries, labor inductions, and episiotomies (incisions of the tissue near the vagina)—although not C-sections—were standard.
With the new feminism of the 1970s, women began demanding more natural births, in which they felt they had more control and were treated with more respect. They wanted “empowering” and “empowered” experiences. In the late 1960s, Zoe Resch’s mother, Mimi Zoet Cummings ’63, had visited an obstetrician early in her first pregnancy. Afterward the doctor had said, “You go home and take care of your knitting. I’ll take care of the baby.”
The pregnancy, it happens, miscarried. By 1970, for her pregnancy with Zoe, Cummings had found a new obstetrician who she felt would be more respectful. Having agency herself seemed “so much more sensible.” She insisted on giving birth without anesthesia. She wanted Zoe’s father allowed in the room with her, and was determined to breast-feed after delivery. She prevailed in all cases.
Meanwhile, the medical profession has increasingly sought to standardize care. Hospitals have come to require procedures that minimize the worst outcomes and can be easily regulated. Certain procedures, such as forceps deliveries, have waned because they are difficult to teach and perform. CFM has become established for overseeing and regulating care. And cesareans have increased because in some cases they were essential for preventing the worst outcomes, because they followed other interventions, were relatively easy to teach and perform, and were unlikely to provoke lawsuits.
“A balance needs to be reached,” says pediatrician Ana Langer, professor of the practice of public health and coordinator of the dean’s Special Initiative in Women and Health—“that will allow women to have normal deliveries with as little intervention as possible, and at the same time will be ready to address any unexpected emergencies.” She, Ecker, Osborne, Singer, Lieberman—all agree that reaching such a balance is in part a matter of public and professional education. Says Langer, “Doctors, women, and families should know that an unnecessary C-section does involve increased risk.”
Ecker believes that obstetrics needs to move ever closer to “evidence-based medicine”—the study of risks and benefits, and the application of this knowledge to medical decisions, professional standards, and training. Doctors and institutions should help patients understand risks and the tests, during pregnancy and labor, that measure them. Whenever appropriate, he says, physicians and patients should avoid interventions and prevent that “cascade.” For example, he says, doctors and hospitals should encourage trials of labor after cesareans.
He also declares his respect for midwifery: “If you have a low-risk population” of pregnant women, he says, a “great model” might be “to have midwives providing uncomplicated prenatal care and doing all the uncomplicated deliveries,” while a few doctors focus on problems and perform C-sections. Mount Auburn Hospital in Cambridge has elements of such a model: an active midwife practice attends 38 percent of labors and deliveries. The hospital’s overall cesarean rate in 2012 was 21 percent, the midwives’ 18 percent. In a small but growing program, its midwives also help teach Harvard Medical School students, who witness normal, uncomplicated births—a rare and valuable experience in an education that focuses on what could go wrong, says HMS lecturer Phyllis Gorman, co-director of Mount Auburn’s midwifery service.
In the broadest sense, attaining an optimal C-section rate may be a matter of finding a middle ground between two approaches to birth and risk—between vigilance toward the “disaster waiting to happen” and support for the “physiologically sound process.” That way, surgery happens when necessary, but is avoided in the many cases when it’s not.