Numerous studies show that caloric-intake restriction (which is known to slow aging in mammals) and exercise have dramatic, beneficial effects on mental acuity and motor ability, says professor of molecular and cellular biology Joshua Sanes, director of the Center for Brain Science. But just how these changes improved functioning was unclear. Now Sanes and a colleague, Knowles professor of molecular and cellular biology Jeff Lichtman, have made a discovery that pinpoints mitigation of age-related structural degradation in neural synapses as the reason for this improvement. According to their research, published this August in the journal PNAS (Proceedings of the National Academy of Sciences), exercise and caloric restriction in lab mice visibly protect and rejuvenate synapses, the neural connections that are critical to cognitive and motor function.
Synapses are junctions between two nerve cells; electrical signals travel along nerves and are converted to chemical signals that jump a narrow cleft between the sending and receiving parts of the synapse. Unlike most cells, says Sanes, neurons aren’t replaced; they tend to be stable throughout life. But with age, a synapse undergoes regressive structural changes--“little pieces of it fragment, or disappear, or the nerve may retract from it; in some cases, the synapse gets so bad that it actually disassembles.” Such degradation, Sanes explains, “interferes with transmission of information from nerve to muscle and leads to the muscle wasting that accompanies old age.”
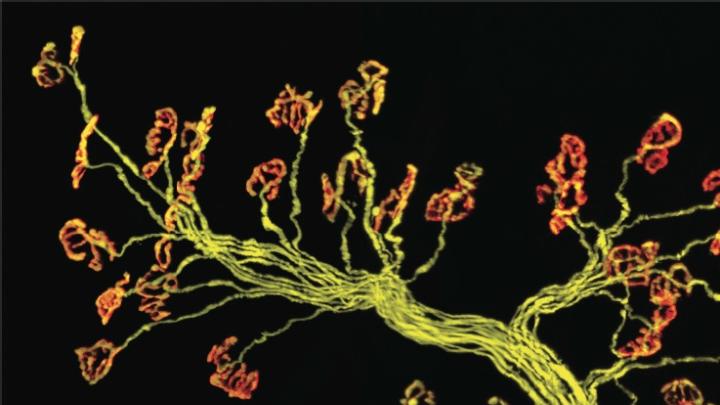
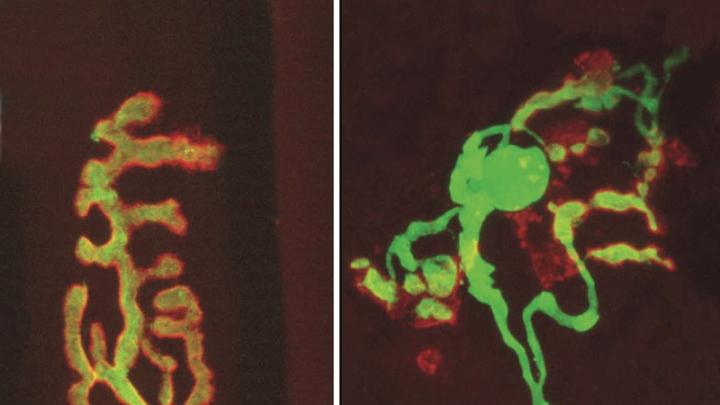
Key to the experiment was a technique developed by Sanes and Lichtman that allows individual synapses to be tagged with a fluorescent protein. (The researchers studied neuromuscular junctions in skeletal muscle, rather than synapses in the brain, because the former are large enough to be seen using a light microscope.) The technique allows investigators to surgically open, and capture an image of, a single tiny synapse, and then find the same synapse later, during a second surgical procedure to observe and document the effects of different protocols.
Sanes and Lichtman found that reducing caloric intake in mice by 40 percent for 24 months protected both muscle fiber and motor neurons (which diminish in number with age), as well as synapses. Their exercise experiment gave the lab mice, which typically live two to three years, an exercise wheel at 22 months of age (“Unlike humans, mice actually run voluntarily,” notes Sanes). Just one month later, they observed in the synapses that they had surveyed earlier “a partial reversal of structural alterations that had already occurred.”
Because the exercise and caloric-restriction experiments were of different duration, Sanes and Lichtman will now reverse the conditions, testing lifelong exercise against short-term caloric reduction. Meanwhile, one of their postdoctoral fellows is conducting similar experiments, this time using synapses in the brain. “The research gives us a hint,” says Sanes, “that the way these extremely powerful lifestyle factors act is by attenuating or reversing the decline in our synapses.”