Main Menu · Search ·Current Issue ·Contact ·Archives ·Centennial ·Letters to the Editor ·FAQs
  |
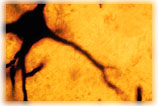 In REM sleep, large brainstem neurons like the one shown above fire sustained bursts of impulses that excite much of the brain. These impulses, also associated with the rapid eye movements that characterize REM sleep, have been recorded on the large display to the left. In REM sleep, large brainstem neurons like the one shown above fire sustained bursts of impulses that excite much of the brain. These impulses, also associated with the rapid eye movements that characterize REM sleep, have been recorded on the large display to the left. |
The dreams most of us remember tend to be action-packed, emotional, and strange. The dreamer is often falling, escaping, or insecure. I recall a dream where I was in a wide sunken walkway or trench with many other people. A lot of pink globes were in the sky, and the mood was festive. All at once I heard an odd sound, looked up, and saw all the pink globes racing toward a distant Cinderella-style castle to my right. I was no longer in the trench but on a green field where the ground sloped upward toward the castle. Many people, perhaps those from the trench, were in the field, and we were all moving toward the castle. I feared what might happen next. I never found out, because just then I awoke. But like innumerable other dreamers, I was impressed by my dream's zany orientation and perplexed as to its cause.
Awhile ago I learned a bit about dreaming from a readable little book called The Chemistry of Conscious States that was written by a man named Hobson. Until then I had considered dreams enigmas. Indeed, the cause of dreams seemed fully as mysterious to me as the movement of certain stars must have seemed to medieval astrologers before Copernicus discovered that the earth and planets revolve about the sun. But of course we all love mysteries, a fact that goes a long way toward explaining why, throughout history, we have embraced the mystery of dreams, hiring untold generations of prophets and soothsayers, and more recent generations of psychoanalysts and dream interpreters, to probe the shadows and explain what our dreams really mean.
True knowledge of what causes our nocturnal imaginings has been a long time coming. For most of the twentieth century, the best we could do was agree or disagree with the 1900 dream theory of Sigmund Freud. Freud said, in essence, that dreams arise from a troubled subconscious, contain forbidden wishes we can't bear to acknowledge, and seem bizarre or choppy because a mental censor disguises or removes the forbidden thoughts.
Just where Freud's influential theory came from is unclear. It could have come from the flawed, rudimentary neuroscience of the 1890s, because Freud had studied that; or else, as Freud claimed, he may have developed it by ruthlessly probing his own emotions and earliest memories. Whatever the case, Freud was driven to develop his hypothesis by an awareness of strong similarities between dreams and certain kinds of madness, and by the realization that one might help a lot of people if one could penetrate to the heart of the mystery. Unfortunately, Freud's theory may have been precipitous: although it provided a rich base for development of psychoanalysis, its lack of clear scientific roots presented a slippery slope for those wishing to probe its scientific merit or devise alternative dream theories of their own.
In Freud's day, of course, scientific study of the mind was limited. Natural science was simply not up to revealing the mind's secrets, even though groundwork for later progress was being laid; and resignation on this score finally froze into the pessimistic doctrine of behaviorism--which said science had no business studying mental events and should limit itself to how humans and animals behave.
 A dream and its investigators in a painting by Paul Delvaux (L'Ecole des Savants, Vienna Museum of Modern Art). The dream itself appears at center-stage, while a psychoanalyst observes a patient on the right and a neurologist examines a dead brain on the left. The painting suggests Delvaux was not optimistic about either approach, preferring to celebrate rather than resolve the central mystery. Painting by Paul Delvaux; MUSEUM MODERNER KUNST, STIFTUNG LUDWIG, VIENNA A dream and its investigators in a painting by Paul Delvaux (L'Ecole des Savants, Vienna Museum of Modern Art). The dream itself appears at center-stage, while a psychoanalyst observes a patient on the right and a neurologist examines a dead brain on the left. The painting suggests Delvaux was not optimistic about either approach, preferring to celebrate rather than resolve the central mystery. Painting by Paul Delvaux; MUSEUM MODERNER KUNST, STIFTUNG LUDWIG, VIENNA |
Even today, remnants of this old pessimism linger, which makes the actual progress to date all the more startling. In fact, especially since the 1950s, we have learned volumes about the human brain--how its roughly 100 billion neurons (nerve cells) receive, process, and transmit messages; how groups of neurons interact; how structures within the brain work with one another; and how the brain's chemistry influences its mental state. Indeed, aided by powerful imaging tools that allow us to penetrate the skull's bony x-ray barrier and watch the mind at work, we now confront a fast-moving river of knowledge that shows promise of unraveling much of the mystery of dreams, learning, memory, thought, and even human consciousness itself.
One powerful tributary feeding this great torrent has been biological research on sleep and dreams. Harvard first fostered such research in the 1960s, when it let an unorthodox psychiatry resident named J. Allan Hobson, M.D. '59, split his time between doing animal research at the medical school's physiology department and performing his normal residency duties at the nearby Harvard-affiliated Massachusetts Mental Health Center. In 1968, after completing his residency and getting extensive training in neurobiology, Hobson got a grant from the National Institutes of Health for animal studies, and the head of Harvard's psychiatry department, Bullard professor Jack Ewalt, allocated some of his own discretionary research funds to buy his young colleague equipment. Hobson thereupon created Harvard's Laboratory of Neurophysiology, which he still directs.
Although Hobson's real goal was to learn about the mind, his first research target was REM sleep, the special sleep phase in humans and mammals characterized by "rapid eye movement." Discovered in the 1950s, REM sleep had been associated with rapid heartbeat, fast breathing, special brain-wave patterns, high levels of brain activity, and vivid dreaming. Typically, a sleeping person's brain passes through various nonREM sleep stages, during which its general level of activity decreases, before entering the very active REM stage--a cycle that repeats itself about every 90 minutes. In humans, dream reports indicate that REM sleep dreams are much more likely to be colorful and imaginative, while those occurring at other times tend to be highly repetitive and dull.
Hobson knew from experience that cats were useful subjects for REM sleep research. As he explained later, in The Chemistry of Conscious States, "The cat's sleep pattern is strikingly similar to our own. Cats' brain waves, eye movements, and muscle twitches clearly differentiate waking, nonREM, and REM sleep in a manner that is parallel to what people experience." Also, REM sleep seemed to be controlled from the brainstem, an ancient part of the brain, just above the spinal cord, that humans share with all vertebrates. So Hobson decided to probe cat brainstems with microelectrodes in order to detect the firing of individual brainstem neurons and get a better idea of what was happening.
 We have learned much about human sleep from cats, whose sleep patterns resemble ours. Time-lapse pictures like these from the Dreamstage exhibit suggested to Hobson that sleepers change position mostly when entering or leaving REM sleep--an idea that led him to develop a sleep-charting device called the "Nightcap" (see photo, lower right). TED SPAGNA-PHOTO RESEARCHERS, INC. We have learned much about human sleep from cats, whose sleep patterns resemble ours. Time-lapse pictures like these from the Dreamstage exhibit suggested to Hobson that sleepers change position mostly when entering or leaving REM sleep--an idea that led him to develop a sleep-charting device called the "Nightcap" (see photo, lower right). TED SPAGNA-PHOTO RESEARCHERS, INC. |
Around this time he was joined by Robert McCarley '59, M.D. '64, a medical resident with a love of neurobiology and a bent for computer programming who met Hobson through one of the latter's seminars, helped him set up the Neurophysiology Laboratory, and thereafter worked closely with him for 16 years. Together they figured out how to immobilize the brain and how to use a grid system to determine where the probe was at any time. They confirmed that their tiny probes could indeed record single nerve cells firing, while the observed cat slept naturally; and by sending the electrical signals to both an audio system and a visual recorder, they could hear these discharges as well as see them. They also confirmed that different nerve cell groups within the brainstem, and elsewhere in the brain, sounded different; and so, by moving from a known group of neurons to some other group, they could proceed to map the brain.
But mapping was not their goal. They wanted to know how the brainstem controlled REM sleep, for which purpose they could draw on a growing body of information. Among other things, they knew the brainstem was a major source of two brain chemicals, norepinephrine and serotonin. They knew these chemicals regulated or slowed the firing rates of certain neurons, played crucial roles in a number of basic brain functions like judgment and learning, and helped govern the entire brain's mental state. They also knew REM sleep was related to bursts of nerve impulses coming up from the brainstem that seemed to excite large parts of the brain. However, the only brain chemical known to cause REM sleep was neither norepinephrine nor serotonin, but rather acetylcholine, which encouraged many of the brain's nerve cells to fire faster. Thus, the exact role of these various brainstem chemicals in generating REM sleep was unclear.
One day in 1973, while exploring a brainstem in Hobson's lab, a researcher named Peter Wyzinski found something interesting. As Hobson relates in another of his books, The Dreaming Brain, "Peter Wyzinski called me into the recording room to observe a faintly visible, slowly discharging cell on the oscilloscope screen....The cell...was...unique in our experience because it stopped firing during REM sleep: it was the first such cell we had ever seen....I instructed Wyzinski not to move the electrode but rather to record this anomalous cell as long as possible. In repeated REM sleep episodes, we found that the cell invariably showed progressive rate decreases during nonREM sleep to a nadir of activity early in the REM period."
 A sleep volunteer wearing the Nightcap. A sleep volunteer wearing the Nightcap. |
In other words, here was a brainstem neuron that did not burst into frenzied activity like many of its companions when REM sleep started. Rather, it slowed its firing rate in REM sleep to almost nothing. This suggested that there were two distinct groups of neurons in the brainstem relating differently to REM sleep--one that turned on around the start of REM sleep and another that turned off.
Later examination showed that Wyzinski's microelectrode had been a hair off the mark. Instead of being at its intended place in the brainstem, where neurons typically turn on in REM sleep, it was in what scientists call the "locus ceruleus," the main area where brainstem neurons take norepinephrine and send it through their long axons to other parts of the brain. More research showed that most neurons in this area turned off as REM sleep started, and so did brainstem neurons elsewhere that distributed either norepinephrine or serotonin.
Working with the information this discovery provided, Hobson and McCarley became convinced that a simple brainstem mechanism caused REM sleep. According to their theory, when a person is awake, brainstem neurons releasing norepinephrine and serotonin suppress those brainstem neurons that distribute acetylcholine, thus dampening down the brainstem's acetylcholine system. But in sleep the brain's general level of activity falls; and after passing through various sleep phases the norepinephrine-serotonin neurons get so inactive that they no longer suppress the acetylcholine generators, unleashing the brainstem's acetylcholine system like a genie released from its lamp. Of course, this genie does not grant wishes. Instead, it sends bursts of acetylcholine through the brain, evoking REM sleep by exciting visual, motor, emotional, and other centers; raising the sleeping brain's general level of arousal; and incidentally sending motor signals to the small muscles of the eyes.
The sleeper doesn't waken because the acetylcholine-driven brain is operating quite differently from when the sleeper is awake: norepinephrine and serotonin are at a low ebb; and the circuits that carry most incoming (sensory) and outgoing (motor) impulses have been blocked by the brainstem, temporarily anesthetizing and paralyzing the sleeper. However, the process is self-limiting. By releasing acetylcholine, the genie excites much of the brain--including the temporarily inactive brainstem cells that had kept it suppressed; and these cells, upon reactivation, release enough norepinephrine and serotonin to suppress it once again. With the genie thus confined, the stimulating rush of acetylcholine eases, the brain's level of activity falls, external circuits unblock, and REM sleep gives way to less exotic rest. In this way the sleeper proceeds, in roughly 90-minute cycles through the night, until waking is induced by a familiar panoply of people, alarm clocks, dawn, the internal prompting of daily (circadian) rhythms, and the complex restorative effects of sleep.
This idea, which Hobson and McCarley called the "reciprocal interaction hypothesis," showed how the brainstem could turn REM sleep on and off, and how REM and other sleep stages could result from a rhythmic chemical dance within the brain. It accounted for why the brain is so active in the REM period without waking, because the chemistry of REM sleep and the waking state are so different. And it explained why judgment and memory seem to work poorly in REM sleep, because the serotonin and norepinephrine shown by other researchers to be necessary for good judgment and memory are lacking.
Beyond that, this concept shed a powerful light on what caused REM dreams--the vivid, bizarre, and colorful dreams that provide much of the fuel for psychoanalysis. Among other things, it indicated that dreams, like much of the brain, are activated in REM sleep by floods of nerve impulses coming up from the brainstem that release the stimulant acetylcholine. The typical REM-sleep dream is strange because this process calls forth choppy, chaotic bits of internal information that are poorly "synthesized" or stitched together. The dream registers in the dreamer's awareness because the brain is highly active at the time. The dream gets accepted uncritically as reflecting reality because the dreamer's critical faculties lack the serotonin and norepinephrine they need for proper judgment. And the dream is mostly or entirely forgotten because this same chemical shortfall disables memory. The new theory implied that in contrast to what Freud had suggested, dreaming is more akin to a regular, internally generated delirium than it is to neurosis.